|
At postnatal stages, the bone marrow contains a mixture of cells, including hematopoietic stem cells (HSCs), adipose cells and fibroblasts, with MSCs making up 0.001-0.01% of the total population. 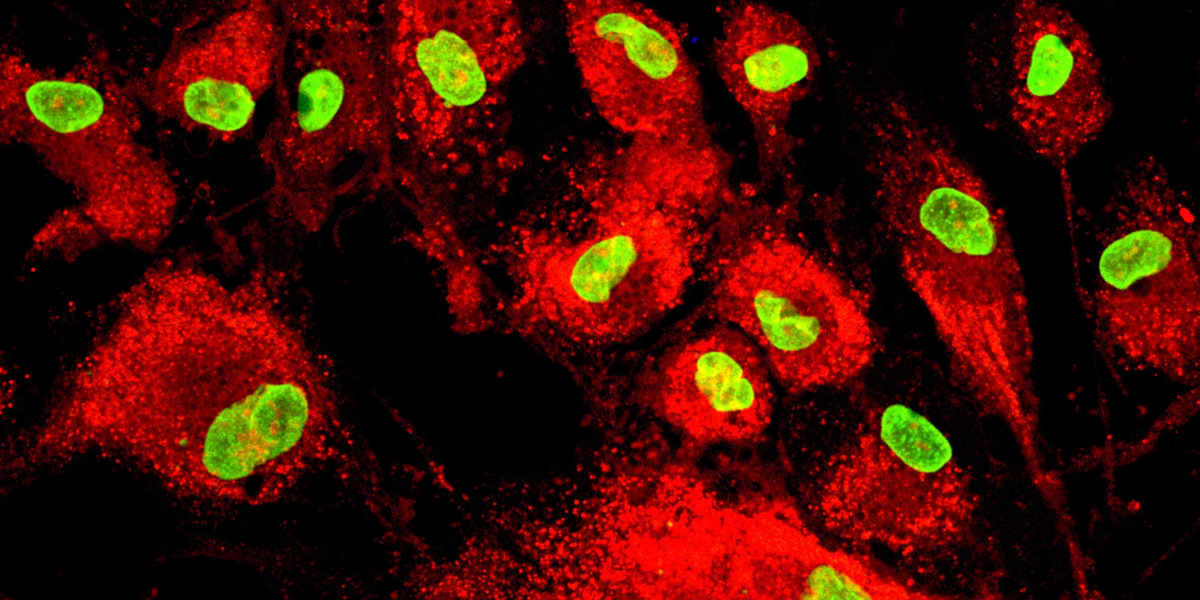
These cells, often referred to as bone marrow stroma cells, develop during bone formation, from the early osteoprogenitor cells which later invade along the forming blood vessel and enter the bone marrow. In this regard, comprehensive cell lineage tracking experiments will significantly promote our current knowledge regarding the in vivo origin and function of MSCs.īone marrow was the first MSC source to be discovered. Moreover, other embryonic or adult MSC sources have been postulated (e.g., neuroectoderm origin), however, their in vivo characterization or contribution to the overall MSC pool is poorly understood. In this regard, known markers such as NT5E (CD73), ENG (endoglin CD105), THY1 (CD90) are shared by both in vitro and in vivo MSCs, however other annotated markers such as STRO-1, D7-FIB, MSCA-1 (W8B2), NGFR (CD271) appear to be rather specific to in vivo MSCs. In our attempts to map the embryonic and adult origin of MSCs, several sources (i.e., tissues and organs) shown to contain presumptive MSCs, characterized by known and rather newly identified markers, have been annotated in LifeMap Discovery. However, some MSCs may originate in other cell niches. Recent publications support the hypothesis that MSCs develop from the blood vessel wall and belong to a subset of perivascular cells (i.e., pericytes). Several fetal and adult MSC sources have been identified so far, however genuine proof for the ability of the derived cells to function as MSCs have been postulated mainly by their phenotypic characterization in vitro. When derived from fetal membranes, such as chorionic and amniotic membranes, MSCs are considered an intermediate between human embryonic stem cells (hESCs) and adult stem cells. MSCs are considered adult or somatic stem cells and remain in a non-proliferative, quiescent state during most of their lifetime, until stimulated by the signals triggered by tissue renewal, damage and remodeling processes. 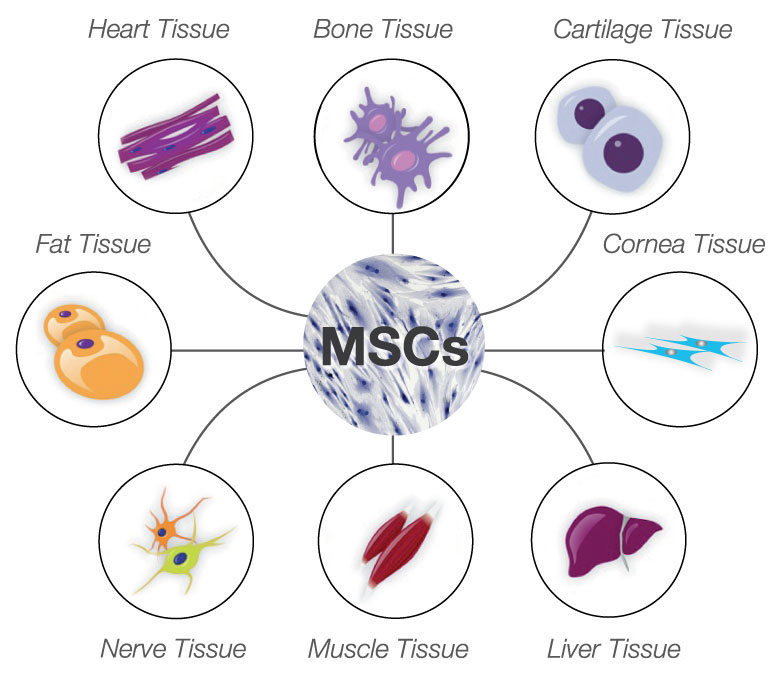
Like other stem cells which reside in pre-natal, adult tissues and organs, mesenchymal stem cells (MSCs also known as multipotent mesenchymal stromal cells) are capable of self-renewal and multipotential differentiation. Embryonic and Postnatal Development of Mesenchymal Stem Cells:
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
